CLL Research – ALPINE Study Compares Zanubrutinib to Ibrutinib in Relapsed/Refractory CLL or SLL
At the recently concluded European Hematology Association (EHA) 2021 Virtual Congress, Dr. Peter Hillmen, MB ChB, Ph.D., gave an oral presentation during the Presidential Symposium entitled: First Interim Analysis of ALPINE Study: Results of a Phase 3 Randomized Study of Zanubrutinib vs. Ibrutinib in Patients with Relapsed/Refractory Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma.
Professor Hillmen is a consultant hematologist (CLL specialist) at Saint James University Hospital (National Health Service) and a professor of Medicine at the University of Leeds in the United Kingdom. Professor Hillmen is a world-renowned researcher and clinician in lymphoproliferative diseases, including CLL. He has published over 80 peer-reviewed papers in journals including The Lancet, New England Journal of Medicine, Blood, and Journal of Clinical Oncology, has authored numerous book chapters, and was Associate Editor of the Haematological Journal.

Background – ALPINE Study Compares Zanubrutinib to Ibrutinib
The treatment of chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL) transformed with the advent of effective inhibitors of B-cell receptor (BCR) signaling. BCR signaling plays a significant role in B-cell proliferation (rapid growth), migration (movement from bone marrow to lymph nodes and blood), and adhesion (cells sticking together). There are two classes of drugs that inhibit the BCR pathway. The first class is the Bruton’s Tyrosine Kinase (BTK) inhibitors which include, ibrutinib (Imbruvica®) and acalabrutinib (Calquence®), which are currently FDA-approved for treating CLL. Zanubrutinib (Brukinsa™) is approved for mantle cell lymphoma (MCL) and is awaiting approval for CLL. Another promising BTK inhibitor is pirtobrutinib, formerly known as LOXO-305.
The ALPINE study is the first clinical trial that compares zanubrutinib to ibrutinib in a head-to-head comparison.

The second class of drugs that inhibit the BCR pathway are the PI3k inhibitors that include idelalisib (Zydelig®), duvelisib (Copiktra®), and the investigational drug umbralisib (UKONIQ™). These agents are very potent inhibitors of B-cell proliferation; however, their significant side effects limit their use at this time.
Ibrutinib was the first targeted therapy for CLL approved by the FDA in 2014 for relapsed or refractory CLL. Ibrutinib, a once-daily oral BTK inhibitor, is the only targeted therapy to demonstrate both a significant progression-free survival (PFS) and overall survival (OS) benefit in multiple randomized phase III studies in both treatment-naïve (TN) and relapsed or refractory (R/R) CLL/SLL. Ibrutinib is effective in controlling CLL progression alone or in combination for approximately 80 percent of patients. However, side effects such as atrial fibrillation and rises in blood pressure have caused some patients to stop taking the medication. Ibrutinib, the first-generation BTK inhibitor, is a standard of care for CLL/SLL.
Zanubrutinib is a second-generation BTK inhibitor, as is acalabrutinib. Zanubrutinib (BGB-3111) is an investigational, second-generation BTK inhibitor designed to maximize BTK occupancy and minimize off-target inhibition of TEC- and EGFR-family kinases. Increased specificity may minimize toxicities reported with ibrutinib potentially due to off-target inhibition such as diarrhea, thrombocytopenia, bleeding, atrial fibrillation, rash, and fatigue.
Methods - ALPINE Study Compares Zanubrutinib to Ibrutinib
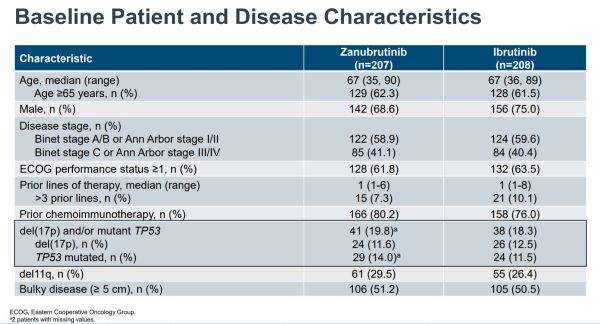
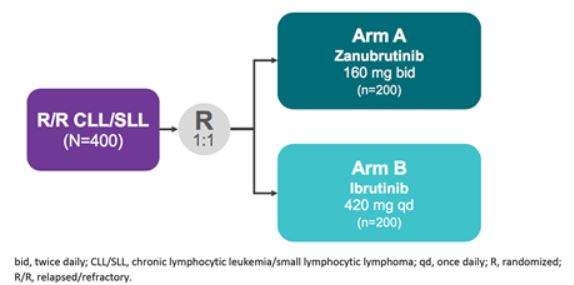
Secondary endpoints were to determine rates of atrial fibrillation, duration of response, progression-free survival (PFS), overall survival (OS), time to treatment failure, PR with lymphocytosis or higher, patient-reported outcomes, and safety.
Results – Interim Analysis of Phase III ALPINE STUDY
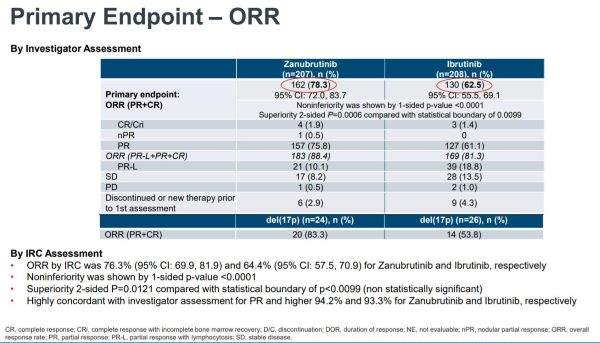
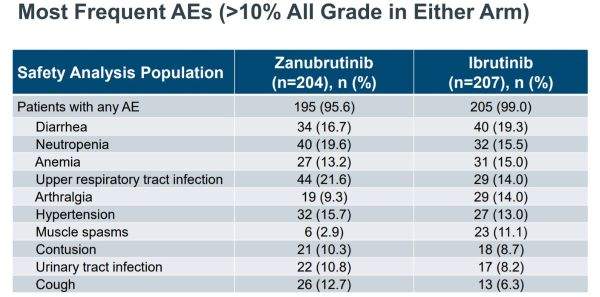
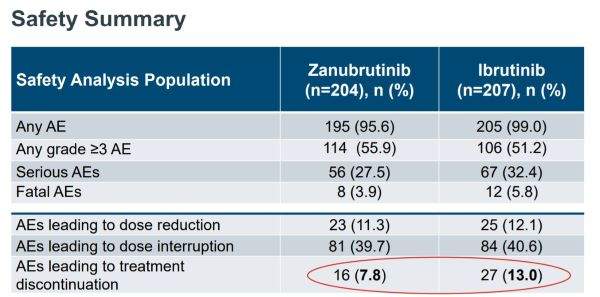
Conclusions – Zanubrutinib Shows Early Promise in CLL/SLL
Ibrutinib now has data for patients after as much as seven years after entering the Resonate-2 study. This long-term data showed less than 50% of patients had disease progression, and a significant number remained on ibrutinib. This longer-term data may limit the early adoption of zanabrutinib as a replacement for the first-generation BTK inhibitor. As clinical data matures, clinicians may decide to use zanabrutinib more widely; until then, Brukinsa may find usefulness in patients at risk of cardiac adverse events or who cannot tolerate ibrutinib.
The ALPINE study is a study that CLL Pharmacist will be looking forward to covering newer data at future conferences.
>



I am currently treating with Acalabrutinib. Are there studies comparing these? Is the cost structure similar?
Michael,
Zanubrutinib is not approved yet for CLL, so I do not have exact costs, but it is in the $14,000 a month range for the cost to the pharmacy. I would stay on acalabrutinib if it works for you because there is more long-term data on acalabrutinib. This data is only about a one-year follow-up.
Tom